Implantable medical device for lymphedema treatment
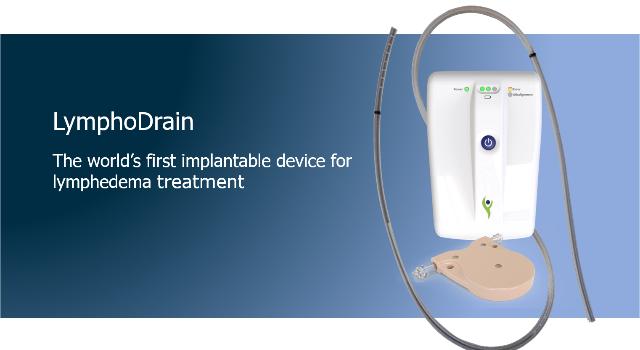
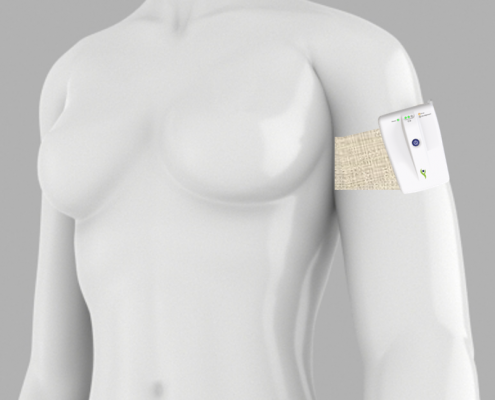
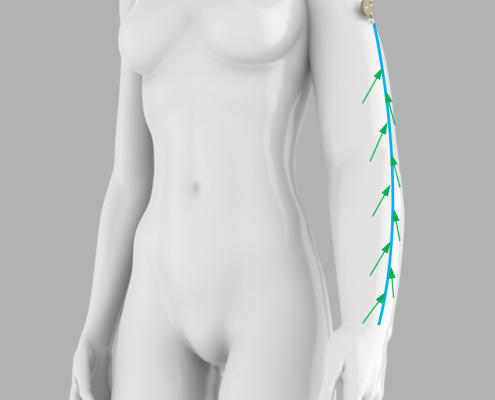
Lymphatica Medtech SA is a young start-up and is developing a minimally invasive product for the treatment of lymphedema. This implantable drainage system with an external controller is the first of its kind. When compared to conventional treatments, the first advantage is that it is a mobile system and can be easily integrated into a patient’s everyday life. Secondly, the overall treatment costs can be lowered because there is less maintenance and the need for complicated individual treatments is reduced. For many patients with lymphedema, it could mean extensive relief in managing their symptoms.
Implant, controller and two mobile applications with cloud connection
konplan developed two mobile apps for convenient operation of the controller and the implant. One app enables doctors to easily adapt and manage therapy plans for their patients and to monitor the controller status. The other app gives patients an overview of their therapy history and all associated data. In addition, data access rights can also be managed in the app, which greatly influences acceptance and the building of trust. The apps are connected to a cloud system for data backup and synchronization, which gives Lymphatica a centralized system for administration and support purposes.
Successful usability study
konplan developed the mobile app and the cloud connection from a single source. The system was successfully tested and evaluated in a usability study. By controlling system parameters via the cloud connection, different scenarios could be performed in a simple manner. In compliance with IEC 62304, the development was implemented in just a few sprints and included plans from the beginning for expanding the apps productive use at any time without needing changes to the architecture.
Milestones
- Specification and design according to medtech standards
- Mockup version of the mobile applications
- Cloud simulation of the hardware
- Evaluated in a usability study